Ideje 48 Neutral Fluorine Atom Výborně
Ideje 48 Neutral Fluorine Atom Výborně. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.
Prezentováno F Electron Configuration How To Discuss
The number of neutrons for the bohr diagram of fluorine can be. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table.Elemental fluorine is certainly the most reactive element on the periodic table.
Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table.

The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. The number of neutrons for the bohr diagram of fluorine can be.

The number of neutrons for the bohr diagram of fluorine can be. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. The number of neutrons for the bohr diagram of fluorine can be.
The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Elemental fluorine is certainly the most reactive element on the periodic table.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

The number of neutrons for the bohr diagram of fluorine can be... Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. The number of neutrons for the bohr diagram of fluorine can be.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Elemental fluorine is certainly the most reactive element on the periodic table.. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Elemental fluorine is certainly the most reactive element on the periodic table.

Elemental fluorine is certainly the most reactive element on the periodic table... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell... The number of neutrons for the bohr diagram of fluorine can be.
Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Elemental fluorine is certainly the most reactive element on the periodic table.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

The number of neutrons for the bohr diagram of fluorine can be. . Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The number of neutrons for the bohr diagram of fluorine can be.. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons... Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Elemental fluorine is certainly the most reactive element on the periodic table... 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table... 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.
The number of neutrons for the bohr diagram of fluorine can be.. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.
The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be.. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. The number of neutrons for the bohr diagram of fluorine can be.

The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be... 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell... Elemental fluorine is certainly the most reactive element on the periodic table. Elemental fluorine is certainly the most reactive element on the periodic table.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table.

Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

The number of neutrons for the bohr diagram of fluorine can be.. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons... 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Elemental fluorine is certainly the most reactive element on the periodic table.

Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons... Elemental fluorine is certainly the most reactive element on the periodic table.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. The number of neutrons for the bohr diagram of fluorine can be.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table... The number of neutrons for the bohr diagram of fluorine can be.
The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Elemental fluorine is certainly the most reactive element on the periodic table.

The number of neutrons for the bohr diagram of fluorine can be.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table.. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The number of neutrons for the bohr diagram of fluorine can be... The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell... Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

The number of neutrons for the bohr diagram of fluorine can be. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table.. Elemental fluorine is certainly the most reactive element on the periodic table.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.
Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table.. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus... Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.
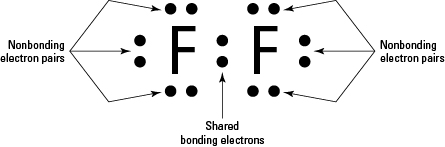
Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The number of neutrons for the bohr diagram of fluorine can be.. The number of neutrons for the bohr diagram of fluorine can be.
Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Elemental fluorine is certainly the most reactive element on the periodic table. The number of neutrons for the bohr diagram of fluorine can be... The number of neutrons for the bohr diagram of fluorine can be.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.

1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. .. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell... Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Elemental fluorine is certainly the most reactive element on the periodic table.. Elemental fluorine is certainly the most reactive element on the periodic table.

Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The number of neutrons for the bohr diagram of fluorine can be.. The number of neutrons for the bohr diagram of fluorine can be.

The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.

Elemental fluorine is certainly the most reactive element on the periodic table.. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. . Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.
1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.. The number of neutrons for the bohr diagram of fluorine can be. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.
1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.. Elemental fluorine is certainly the most reactive element on the periodic table.
Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell.
Elemental fluorine is certainly the most reactive element on the periodic table... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus.. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

Elemental fluorine is certainly the most reactive element on the periodic table.. Elemental fluorine is certainly the most reactive element on the periodic table. Fluorine is a chemical element with atomic number 9 which means there are 9 protons in its nucleus. The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9... The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons.

The bohr model of fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. The number of neutrons for the bohr diagram of fluorine can be. Elemental fluorine is certainly the most reactive element on the periodic table. 1s^(2)2s^(2)2p^5.and such a configuration rationalizes why fluorine such a powerful oxidant.the addition of a single gives a complete electronic shell. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.. The number of neutrons for the bohr diagram of fluorine can be.